The isotope has fourteen neutrons in its nucleus. 
$ The isotopic abundance of is $ 99.9% $. If the thermal evaporator reactor has the planetary geometry with a radius of 40 cm, calculate the deposition rate of A l in unit of m / sec The atomic mass of aluminum is 27 and the mass density of solid Al is 2700 kg / m 3. Thus, the number of protons in the nucleus of an aluminium atom is $ 13. At 1373 K the vapor pressure of aluminum is about 1 × 1 0 3 torr. Also, it has one-third of the density of iron. Furthermore, aluminum allows that it has a relatively lower density than steel. This is known as aluminums embodied energy.In 2014, 49. Moreover, its density is constant but is heavier than that of water. The extraction of aluminum is extremely energy intensive it requires 190-230 megajoules of primary energy per kilogram of aluminum extracted and processed. Also, this means that every 1.0 of aluminum has a mass of 2700 kg. density of aluminium is equal to 2 699 kg/m³ at 20☌ (68☏ or 293. The density of aluminum is about 2700 that is equal to 2.7. $ The number of electrons is always equal to the number of protons so that the atom remains electrically neutral. Aluminium weighs 2.699 gram per cubic centimeter or 2 699 kilogram per cubic meter, i.e. 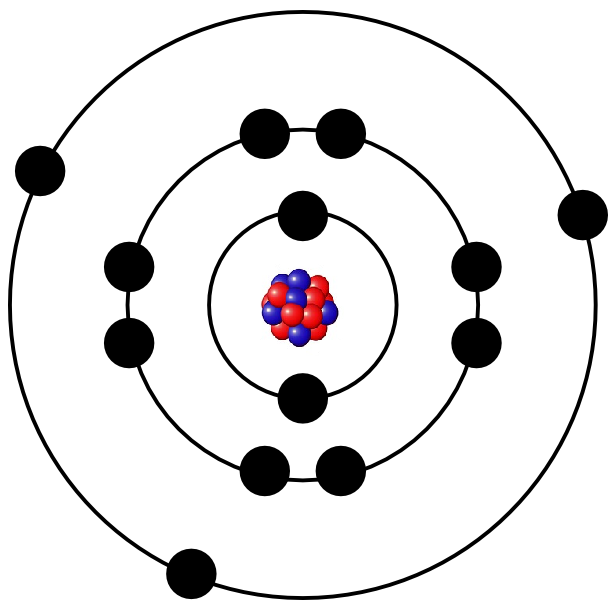
$ Thus, the number of electrons around the nucleus of an aluminium atom is $ 13. $ The mass of an atom of any element is the sum of the atomic number of the element and the number of neutrons. We know that the number of neutrons in the nucleus of the aluminium atom is $ 14. $ Its nucleus thus contains $ 13 $ protons. The atomic number (Z) of aluminum is $ 13. In SI unit (kg) we can say that 1 amu is equal to. Additional information: An atomic unit of mass is defined as accurately. Atomic mass is expressed in the atomic mass unit. The Atomic Mass of aluminium is 26.98 amu. Therefore the atomic mass of aluminium is 27 amu or 27. Hint :We know that the atomic mass is the mass of a single atom of a chemical element is the sum of the atomic number of the element and the number of neutrons present in its nucleus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
